Aortic Dissection Review
What do Lucille Ball, John Ritter and Jonathan Larson (author of Rent) all have in common? Well, yes, they were all involved in the entertainment industry, but they also all have the dubious distinction of having died of complications due to aortic dissection.
Aortic Dissection: Function of the Aorta
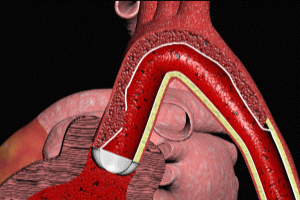
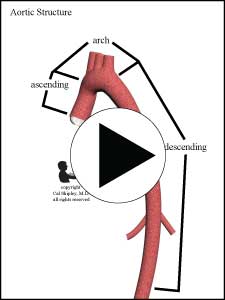
The aorta is the largest artery in the human body. Attached to the left ventricle, it represents the sole means of conveying blood to every organ and tissue in the body, including the heart (via the coronary arteries). As a result, any significant disturbance in blood flow through the aorta can have catastrophic consequences for the affected individual
What Is Aortic Dissection?
The easiest way to understand aortic dissection is to gain an understanding of the structure of the aortic wall. The aortic wall is divided into 3 layers: an inner layer, known as the “intima”, a middle layer known as the “media”, and an outer layer known as the “adventitia”. These 3 layers are present in every artery in the human body, but vary in thickness depending on the size of the vessel. Under normal circumstances, blood is ejected from the left ventricle of the heart into the aorta, and as it flows through the vessel, it comes into contact only with the inner layer, or intima, of the aortic wall. Please refer to the “aortic anatomy” animation following.
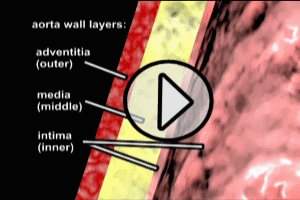
However, in circumstances where there is disease of the intima, or with trauma, a tear may occur. The pressure generated by contraction of the left ventricle pushes blood into the tear, and depending on a variety of factors (size and location of the tear etc.), the blood may push apart the intimal and medial layers. This separation of layers is known as “dissection”, and may occur progressively, with blood flowing either away or towards the heart, dissecting the intimal and medial layers as it goes. Please see the “dissection” animation following to visualize this process.
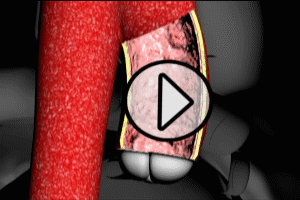
Why Is Aortic Dissection Dangerous?
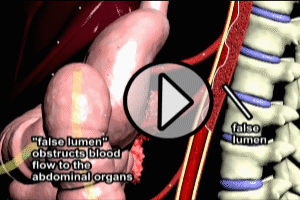
The channel created by blood flowing between the intima and media is known as a “false lumen” (see the “Aortic Dissection” animation above). The creation of a false lumen can have several negative consequences. First, the blood that is flowing into the false lumen, is. by definition, not flowing through the normal aortic lumen, and so flow of blood is reduced to aortic branches (such as the carotid arteries, etc.). As the false lumen enlarges, the reduction in normal blood flow to branch arteries can have serious consequences on organ function (e.g. stroke, heart attack). Second, as the dissection extends along the aorta, the size of the false lumen may be such that it physically blocks the entrances to major arterial branches, severely reducing blood flow to organs. Please see the animation of an obstructive false lumen following.
The process of dissection may involve any of the arterial branches of the aorta, including the coronary arteries of the heart, as well as the aortic valve
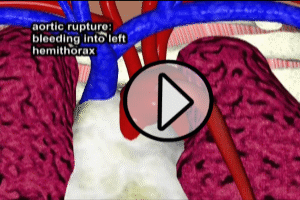
And last, the dissection of layers weakens the overall strength of the aortic wall, and may result in rupture through the wall, resulting in catastrophic internal hemorrhage. If this rupture occurs within the pericardial sac (which contains the heart and the first portion of the aorta) a life-threatening “acute cardiac tamponade” may occur. Please see the cardiac tamponade animation following.

Aortic Dissection: Classification
Aortic dissections are classified as acute or chronic depending on how long the symptoms have been occurring at the time of presentation to a medical provider. Symptoms of 2 weeks or less duration are classified as acute, whereas patients with symptoms of greater than 2 weeks duration are labeled chronic. Typically, serious consequences of aortic dissection, such as obstruction of blood flow to major aortic branches, cardiac tamponade, and aortic rupture, occur within the first 2 weeks after onset of dissection, although it is not uncommon for catastrophic events to occur within minutes of dissection.
Aortic dissection is also classified according to the location of the initial dissection. There are 2 such classification systems in use. The more frequently used Stanford system groups dissections into Type A – those involving the ascending aorta (irrespective of the original intimal tear), or type B, all others. The alternative DeBakey system classifies according to the site of the initial tear, with Type 1 starting with a tear in the ascending aorta and extending to at least the aortic arch, type 2 starting in and confined to the ascending aorta, and type 3 starting in the descending aorta and extending forwards away from the heart (distally) or backwards toward the heart (proximally), with the distal extent above the diaphragm, and 3B, same as 3 but with the distal extent below the diaphragm (see illustrations following).
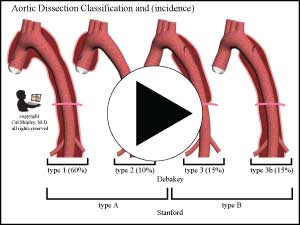
There are some possible variations on the typical scenario of intimal tear followed by dissection, including intimal tear with exposure of the medial la
yer but without dissection of wall layers, and and bleeding within the aortic wall in the absence of a detectable intimal tear. This latter condition, known as Intramural Hematome, or IMH, is generally thought to be a result of spontaneous rupture of tiny blood vessels (known as vaso vasorum) within the medial layer.
Incidence of Aortic Dissection
The incidence of aortic dissection in the general population is about 2-3 out of every 100,000 people, with about 2000 new cases in the US each year. It may affect anyone but is most commonly seen in men in the 60-80 age group. According to the International Registry of Aortic Dissection (IRAD), there is a 3:1 male to female ratio in the occurrence of aortic dissection. However, Aortic dissection is by no means confined to older age groups as a result of several predisposing conditions which may occur in the young.
Risk factors for Aortic Dissection
- Hypertension – the presence of high-blood pressure is the most common predisposing factor in aortic dissection, and has been seen as a causal factor in up to 70% of all cases.
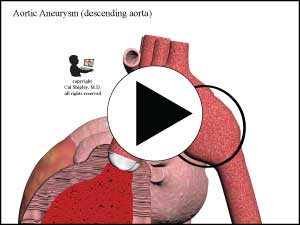
- Preexisting aortic aneurysm – according to IRAD, about 13% of all aortic dissection cases involve the presence of a preexisting aortic aneurysm, and may particularly affect younger patients less than 40 years of age. An aneurysm is a weakened area of the aortic wall –
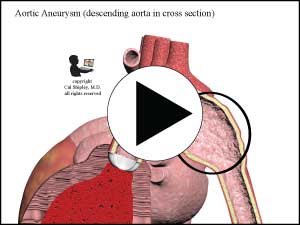
3. Connective Tissue Disorders –such as Marfan’s Syndrome or Ehler-Danlos Syndrome. These may result in aortic dissection in younger patients.
4. Pregnancy and delivery
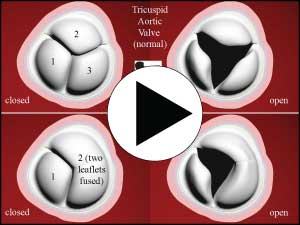
5. Bicuspid aortic valve – is a congenital anatomic abnormality whereby the aortic valve consists of just 2 valve “leaflets” instead of the more common 3 leaflets
6. Inflammatory diseases – such as Giant Cell Arteritis, Rheumatoid arthritis, Syphylitic aortitis, or Takayasu arteritis – all conditions that may cause severe inflammation of, and damage to, arteries, such as the aorta (known as vasculitis)
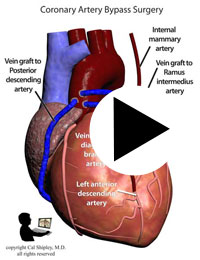
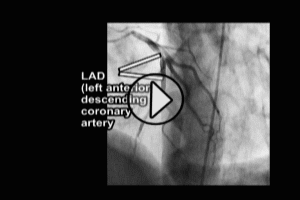
7. Coronary Artery Bypass Graft surgery, Cardiac Catheterization or other procedures involving the use of surgical instruments in the aorta – aortic dissection is a rare complication of these procedures
8. Coarctation of the Aorta – this is a congenital condition in which there is an abnormally narrow section of the ascending aorta, often with a weakened wall structure that can lead to aortic dissection in the young
9. Turner syndrome – Turner syndrome is a genetic chromosomal disorder in which coarctation of the aorta may be a feature. (see 8.)
10. Coarctation of the Aorta – this is a congenital condition in which there is an abnormally narrow section of the ascending aorta, often with a weakened wall structure that can lead to aortic dissection in the young
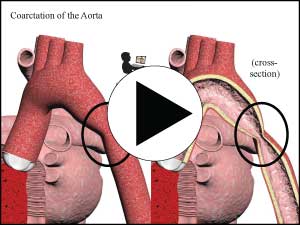
11. Weight lifting – high intensity weight lifting may cause temporary spikes in blood pressure, which van trigger aortic dissection, particularly in those individuals with other preexisiting risk factors for dissection
12. Family history – individuals who have first degree relatives (parents, siblings, or children) with a history of aneurysms or dissections
13. Crack cocaine – crack cocaine use may cause severe increases in blood pressure, which can result in aortic dissection, particularly in individuals with other risk factors.
Symptoms and Signs of Aortic Dissection
Symptoms associated with aortic dissection vary depending on the location and extent of the dissection, and the vascular structures affected. It is of critical importance for the clinician to be aware that the symptoms of aortic dissection may be very vague, with the patient presenting simply with sudden onset of anxiety, or a generalized feeling that something is “not right”. Fully 10% of patients will not experience pain in the early stages of dissection. For this reason, missed diagnosis of aortic dissection early in the course of the condition is unfortunately not rare.
1) Chest Pain – is the most common presenting symptom of aortic dissection, occurring in approximately 90% of acute cases (symptoms of less than 2 weeks duration). Most patients describe the pain as sharp,or knife-like, with an abrupt onset, and unlike any pain they have previously experienced. The classic description of aortic dissection pain as tearing or ripping is less common. Patients with Type A dissections most often complain of anterior (frontal) chest pain, while those with Type B dissections typically experience back and/or abdominal pain. The presence of abdominal pain should raise suspicions for vascular compromise of the blood supply to the gut (mesenteric ischemia)
2) Pulse deficit – refers to a weakening of the pulse noted on palpation in the carotid, brachial or femoral arteries, as a result of obstruction to blood flow by an intimal flap or compression or obstruction by a false lumen (as described above). Pulse deficits may be noted unilaterally or bilaterally (for example, in only one carotid artery or in both) depending on the location and extent of the dissection. Differences in the systolic blood pressure (>20mm Hg) may be observed when comparing the arms. Pulse deficits are more common in patients with aortic dissection involving the aortic arch, or with extensive thoracolumbar (upper and lower aorta) involvement (see thoracolumbar dissection animation here), and far less common with dissections of the descending aorta only. In general, patients with pulse deficits have a poorer prognosis than those without, and are more often hospitalized with serious complications. The presence of carotid pulse deficits is highly correlated with stroke.
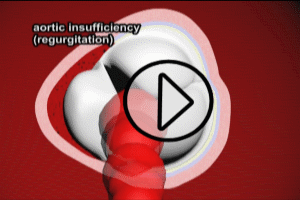
3) Hypotension ( low blood pressure) – along with shock and/or syncope (lightheadedness) are more commonly associated with dissections of the ascending aorta (Type A), whereas hypertension (elevated blood pressure) can be related to dissections of the descending aorta (Type B). Hypotension may be caused by rupture of the aorta (with hemorrhage into the chest or abdominal cavities), cardiac tamponade, aortic valve regurgitation, cardiac ischemia (blood flow impairment in the coronary arteries) or myocardial infarction (heart attack).
3) Syncope (lightheadedness) – may be related to any of the complications listed under “Hypotension”, and may also be related to blood flow impairment to the carotid arteries, reducing flow to the brain. Syncope is more common in patients with Type A dissections.
4) Heart murmur – aortic dissections that extend toward the heart may involve the aortic valve, causing it not to close properly, and allowing blood to flow backwards (regurgitation) across it during the relaxation phase of the cardiac cycle.

5) Neurological deficits – such as limb weakness/numbness or disturbances in vision, hearing or speech, or altered mental status (e.g. confusion, decreased responsiveness or coma) may be caused by dissection-related impairment of blood flow through the carotid arteries (and hence, impairment of oxygen and nutrient flow to the brain). Persistent blood flow impariment to the brain may result in stroke (see my animation here for more information on stroke) or by physical compression of nerves or blood vessels as the aortic wall swells with blood in the false lumen. Examples of compression-related symptoms are vocal hoarseness from recurrent laryngeal nerve compression, Horner’s syndrome from compression of the superior cervical sympathetic ganglia, and paraplegia (sensory or muscular impairment of the lower limbs) due to pressure on the blood vessels supplying the spinal cord.
Diagnosis of Aortic Dissection
Individuals presenting with sudden onset of any of the preceding symptoms, particularly sharp chest or abdominal pain in association with pulse deficits and/or hypotension, and presence of mediastinal widening on chest xray (see “Imaging Studies” below), should be suspected of having aortic dissection.
Physical Exam
Physical examination may yield very little additional information in a patient experiencing an acute aortic dissection, and this lack of findings on exam may direct the medical provider away from the correct diagnosis. In spite of a lack of physical exam findings, it is critical that the examiner maintain a high degree of suspicion based on the presenting symptoms. Nevertheless, there are physical findings that may be found in those experiencing an acute aortic dissection, so an in-depth examination is of paramount importance for those whose symptoms suggest the possibility of an aortic dissection:
- Blood Pressure – may be abnormally low (hypotension), as in Type A dissections, or elevated (hypertension), often seen with Type B dissections. Blood pressures should be carefully recorded in both upper limbs, as dissections may asymmetrically affect blood flow through the brachiocephalic arteries of the aortic arch. Systolic blood pressure differences of more than 20mm Hg are highly suspicious for aortic dissection.
- Asymmetrical pulses related to 1) above.
- Heart Murmurs – new onset heart murmurs, particularly diastolic murmurs heard over the aortic valve due to regurgitation, may indicate valve involvement by dissection.
- Signs of aortic regurgitation – particularly wide pulse pressure and/or bounding pulses.
- Signs of Cardiac Tamponade – muffled heart sounds, hypotension (low blood pressure), pulsus paradoxus, jugular venous distention (abnormal prominence of the jugular veins), or Kussmaul’s sign.
- Neurological deficits – a thorough neurological exam may reveal deficits related to impaired blood flow through the carotid arteries, or related to compression of nerves or vascular structures as discussed in Symptoms and Signs 5) above.
Imaging studies
Chest xray – while a normal plain film AP view of the chest cannot rule out an aortic dissection, the classic finding of widening of the mediastinum may be present. If there has been a rupture of the aorta with hemorrhage into the thorax (chest cavity), signs of hemothorax may be visible.
CT, MRI and TEE (transesophageal echocardiogram) – successful diagnosis of aortic dissection using these methods is dependent on visualization of an intimal flap separating false and true lumens, as well as possible associated complications including: extent of the dissection, thrombus (clot) in the false lumen, dissection in the ascending aorta, dissection of coronary arteries and branch vessels, aortic valve involvement, and pericardial effusion/tamponade
- CT scan – angiography with CT has a high sensitivity and specificity for detecting aortic dissection, and is generally readily available in most emergency departments. Use of spiral (helical) CT appears to improve the accuracy of imaging. The finding of a true and false lumen is critical too diagnosis, as is compression of the true lumen of the aorta. An intimal flap may or may not be seen. CT angiography is less sensitive for detecting dissection in the ascending aorta, and cannot be used in hemodynamically (circulatory) compromised patients due to the need for iodine based contrast material, which can be very toxic to the kidneys.
- MRI – is highly accurate for diagnosing aortic dissection. Use of MRI angiography with Gadolinium contrast has a specificity and sensitivity of 95-100%. Gadolinium is considered to have a better safety profile than the iodine based contrast used in CT scan, however, it may also be toxic to the kidneys in patients with circulatory compromise. MRI may used without contrast, but the accuracy of non-contrast MRI in diagnosing aortic dissection has not been well studied. Other disadvantages of MRI are its decreased availability on an emergency basis at many centers, the time required to perform the exam, the need for patients to remain motionless for up to 30 minutes during an exam, and inability to perform the study in patients with claustrophobia, pacemakers, and certain types of metallic implants.
- TEE (Transesophageal Echocardiogram) – is the preferred imaging study for patients with hemodynamic (circulatory) instability. It requires no contrast material, and is portable, and so can usually be performed within minutes in the Emergency Department. In skilled hands, it has a sensitivity and specificity comparable to that of CT and MRI, and it is most useful in diagnosing dissections involving the ascending aorta. Patients undergoing TEE must have an esophageal intubation performed, which requires some form of sedation. This sedation may be dangerous in some patients with circulatory instability. Physicians and technicians skilled in the use of TEE to accurately diagnosis aortic dissection are essential, and may not be available on an emergency basis. TEE cannot assess aortic dissection below the diaphragm.
Aortography – angiography of the aorta using injections of contrast material was previously the preferred imaging method for diagnosis of aortic dissection, but has largely been replaced by CT, MRI and TEE. However, in patients in whom CT, MRI or TEE findings are inconclusive, or unavailable, digital subtraction angiography is the study of choice.
Electrocardiogram (EKG)
Theoretically, electrocardiogram would be expected to show signs of myocardial ischemia (impaired blood flow to the heart muscle) in individuals where aortic dissection has extended into the coronary arteries. In practice, however, EKG has generally been shown to have low sensitivity in such circumstances.

In patients with dissection related cardiac tamponade, ST segment changes as well as decreased voltage of the QRS complex and a pattern known as “electrical alternans” may be seen. As a rule, no changes are seen in the EKG in patients who do not have coronary artery involvement or cardiac tamponade.
Laboratory findings
Lab findings in aortic dissection are variable and most are very non-specific. Lab changes associated with aortic dissection may include:
- D-Dimer – is a product of fibrin breakdown, which represents activation of the “coagulation cascade”. This occurs when tissue factor (TF) in the aortic media layer is exposed by an intimal tear. D-Dimer elevation may occur in a large variety of conditions, so its presence in the bloodstream in elevated levels is not helpful in making the specific diagnosis of aortic dissection. Levels below 250-500 ng/ml (experts vary on this value), however, are much more useful in eliminating the diagnosis.
- Leukocytosis (increased white blood cell count) – due to physiological stress
- Blood Urea Nitrogen (BUN) and Creatinine levels increased due to renal artery involvement
- Lactate Dehydrogenase (LDH) elevation due to hemolysis (breakdown of red blood cells) in the false lumen
- Creatine Kinase (CK) and Troponin I and T increases due to coronary artery involvement with subsequent myocardial ischemia (impairment of blood flow to the hesrt muscle)
- Smooth Muscle Myosin heavy-chain assay – elevation in first 24 hours highly suspicious for aortic dissection
Cal Shipley, M.D.

